Paper
"Ex Vivo Pilot Study of Cardiac Magnetic Resonance Velocity Mapping for Quantification of Aortic Regurgitation in a Porcine Model in the Presence of a Transcatheter Heart Valve"

Keywords
Transcatheter heart valve | Paravalvular aortic regurgitation | Quantification Cardiac magnetic resonance velocity mapping
Authors
Nynke H. M. Kooistra, Freek Nijhoff, Masieh Abawi, Pierfrancesco Agostoni, Daniël M. Araya Roos, Sjoerd van Tuijl, Niels Blanken, Michiel Voskuil, Pieter A. F. M. Doevendans, Pieter R. Stella, Tim Leiner
Purpose of this study:
"The purpose of this pilot study was to evaluate the accuracy of cardiac MR velocity mapping for the quantification of post- TAVI AR in both balloon-expandable and self-expandable valves and to determine at which anatomic level this analysis is most accurate. To this end, cardiac MR velocity mapping of both antegrade and retrograde flow was used in a porcine cadaver heart model in the presence of different types of pros- thetic THVs."
Abstract
Accuracy of aortic regurgitation (AR) quantification by magnetic resonance (MR) imaging in the presence of a transcatheter heart valve (THV) remains to be established.
We evaluated the accuracy of cardiac MR velocity mapping for quantification of antegrade flow (AF) and retrograde flow (RF) across a THV and the optimal slice position to use in cardiac MR imaging.
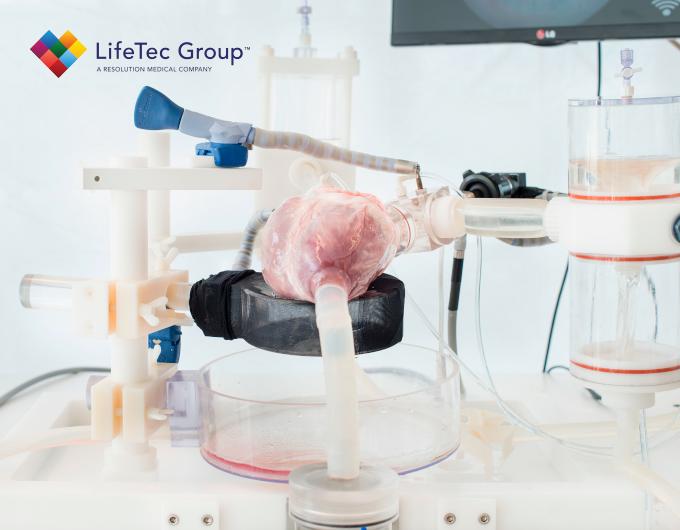
In a systematic and fully controlled laboratory ex vivo setting, two THVs (Edwards SAPIEN XT, Medtronic CoreValve) were tested in a porcine model under steady flow conditions.
Results showed a high level of accuracy and precision. For both THVs, AF was best measured at left ventricular outflow tract level, and RF at ascending aorta level.
At these levels, MR had an excellent repeatability (ICC > 0.99), with a tendency to overestimate (4.6 ± 2.4% to 9.4 ± 7.0%).
Quantification of AR by MR velocity mapping in the presence of a THV was accurate, precise, and repeatable in this pilot study, when corrected for the systematic error and when the best MR slice position was used. Confirmation of these results in future clinical studies would be a step forward in increasing the accuracy of the assessment of paravalvular AR severity.
Transcatheter aortic valve implantation (TAVI) is a well-accepted alternative treatment for patients with symptomatic, severe native aortic valve stenosis who are at high risk for surgical aortic valve replacement (SAVR). TAVI may even outperform SAVR, as demonstrated by lower mortality rates in high-risk patients.
However, aortic regurgitation (AR) after TAVI remains an important challenge. With respect to the incidence and severity of postoperative AR after 30 days, TAVI has a significantly higher rate of moderate or severe AR then SAVR (4.2% vs 0.4%).
Meanwhile, evidence suggests that even lowgrade postoperative AR negatively impacts both functional status and survival after TAVI. AR after TAVI mainly consists of paravalvular leakage, resulting from incomplete circumferential apposition of the prosthetic stent frame to the surrounding calcified tissue. This TAVI-related paravalvular AR is difficult to evaluate by echocardiography, because of acoustic shadowing artifacts originating from the metallic stent frame and the severe calcifications of the native valve that remains in situ. Quantification of paravalvular AR by echocardiography is further complicated by the typical phenotype of this AR, involving multiple, eccentric, and/or wall hugging jets. An emerging novel angiographic method to quantify AR is video-densitometry. Nevertheless, this method is angiography based and thus cannot be used at follow-up.
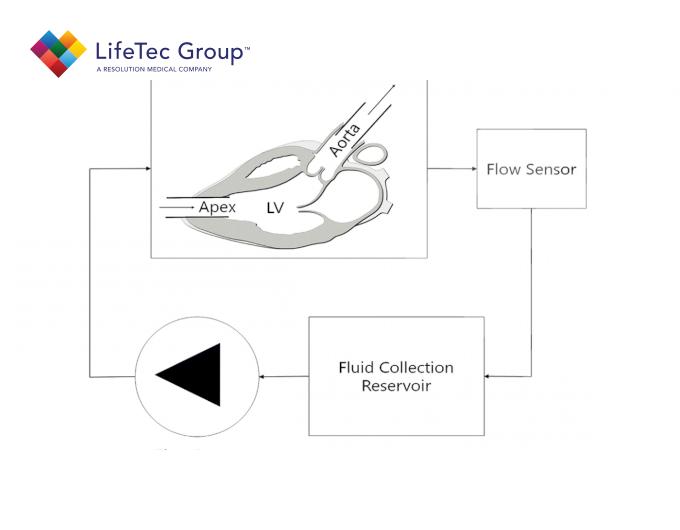
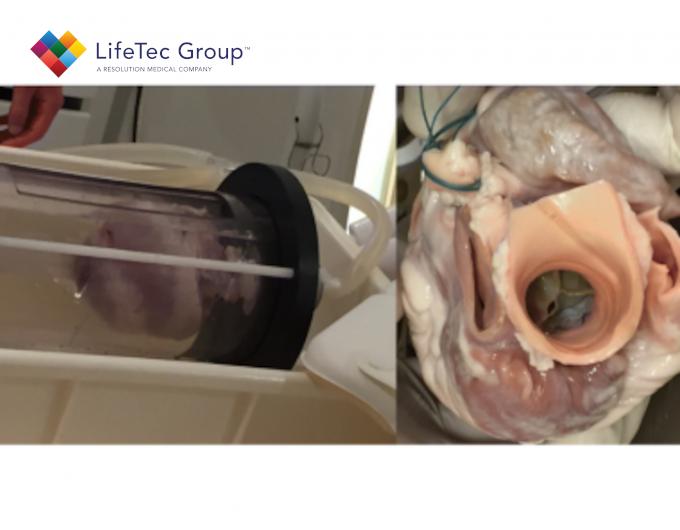
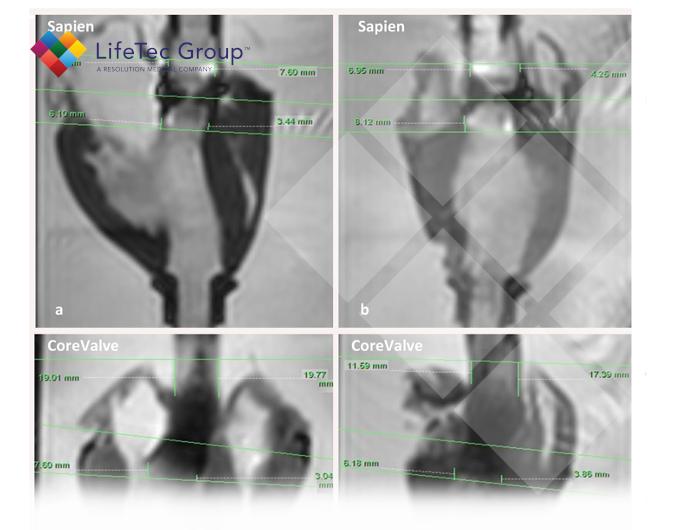
Another promising tool for assessing AR after TAVI more accurately is cardiac magnetic resonance (MR) imaging. Whereas echocardiography mainly relies on qualitative or semi-quantitative measures, and may underestimate or overestimate AR, cardiac MR provides highly accurate quantitative data on regurgitation severity (i.e., regurgitant fraction and volume) with excellent inter-acquisition repeatability and interobserver and intraobserver agreement. However, AR quantification by cardiac MR has only been validated by in vivo and in vitro studies pertaining to AR in a native valve scenario. Reporting on post-TAVI AR evaluated by cardiac MR might therefore be rather speculative, since there is no data on its accuracy in the presence of transcatheter heart valves (THVs). For instance, the metallic stent frame (the backbone of the majority of THVs) could disturb the homogeneity of the magnetic field, which leads to attenuation of the amplitude and errors in the phase of the MR signal. These errors in the phase result in errors of the cardiac MR velocity mapping signal. Moreover, signal void artifacts caused by the stent frame force the imaging slice for through-plane velocity mapping to be placed further away from the zone of the prosthesis-annular apposition, i.e., the location where paravalvular leaks occur, which may lead to underestimation of regurgitation severity. An alternative approach for AR quantification which is less sensitive to stent frame artifacts is calculating the difference between left ventricular and right ventricular stroke volumes based on cine MR imaging. Nevertheless, this is not appropriate in most TAVI patients because of coexisting regurgitation of other heart valves. Currently, convincing data regarding the most appropriate slice position of the cardiac MR velocity mapping imaging in the presence of a THV are lacking. Various positions have been used, varying between the lower border of the stent frame to the level of the ascending aorta. Accuracy of aortic regurgitation quantification by MR imaging in the presence of a transcatheter heart valve remains to be established.
The purpose of this pilot study was...
"The purpose of this pilot study was to evaluate the accuracy of cardiac MR velocity mapping for the quantification of post- TAVI AR in both balloon-expandable and self-expandable valves and to determine at which anatomic level this analysis is most accurate. To this end, cardiac MR velocity mapping of both antegrade and retrograde flow was used in a porcine cadaver heart model in the presence of different types of prosthetic THVs."
Download journal publication:
Interested in more about what we do at LifeTec Group? Contact us!
Call at +31 40 2989393 Or e-mail us
